Cleared combinations of FDA 510 (K) appliances, machines, and materials in the US
The appliance types that are FDA cleared in the US are export models, indirect bonding transfer media and custom metal bands on cleared combinations of production equipment and materials.
Production equipment and materials per appliance type
‘Indirect Bonding Transfer Media’ comes in two types. The traditional one where you print a model for subsequent vacuum pressing:
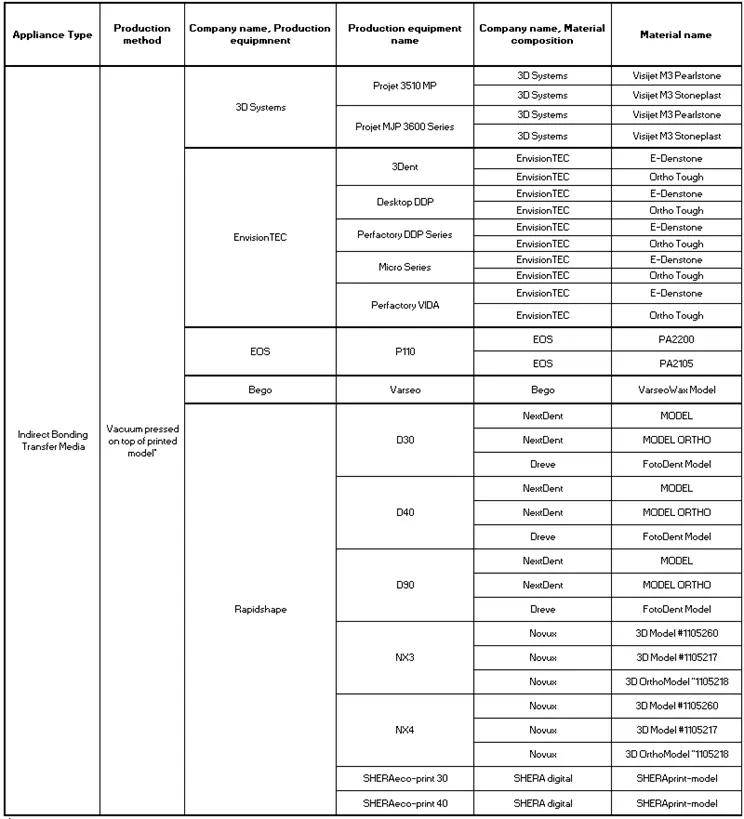
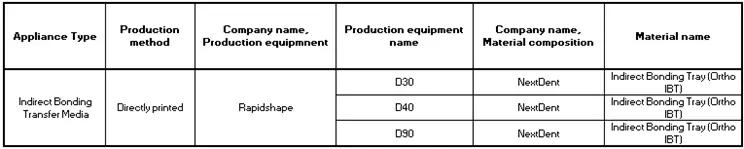
And the new innovative ‘Directly Printed Transfer Trays’ that doesn’t require a model or subsequent vacuum pressing:
‘Export Model’ is the traditionally printed study models or models used for aligner production or other models that might be the basis of further analogue work performed on top of it:
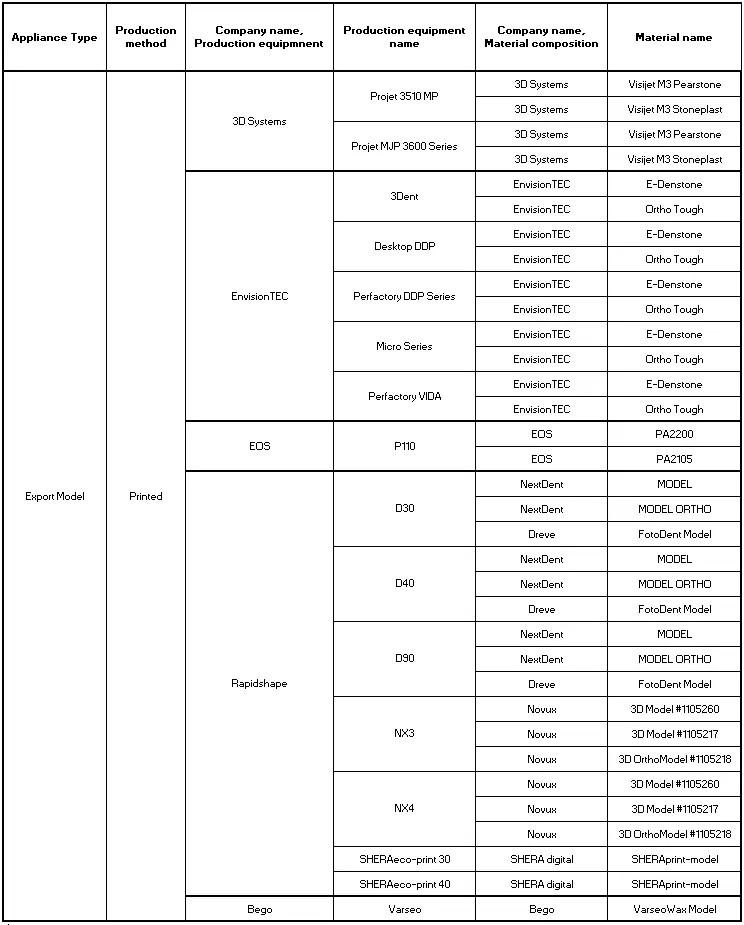
‘Custom Metal Bands’ are used for various appliance that contains its namesake and might be fused later in an analogue workflow with parts created by hand or FDA cleared stock parts:
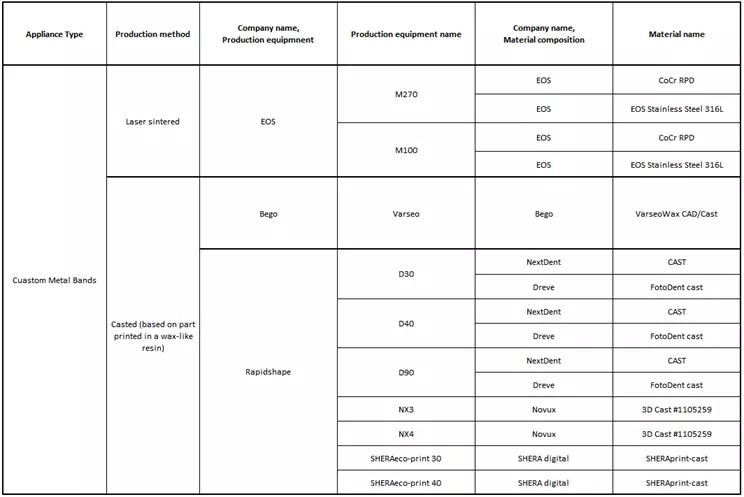
More types pending clearance.